Our Clinical Research 101 series takes an in-depth look at key steps and tips for navigating the clinical research process. The twentieth installment in our Clinical Research 101 series is by Leslie Love, Senior Project Manager at CHÉOS. The following is a brief discussion of source documentation, and why it is important to retain it for the research record.
Ms. Love is a certified clinical research professional with over 25 years of experience in a variety of research settings. Her primary role is to provide project management support to CHÉOS and Canadian HIV Trials Network-supported investigators, as well as develop research training materials and programs. Ms. Love is one of many project managers who work for CHÉOS. The Centre’s project managers are experts in the regulatory, policy, budgeting, and implementation requirements for clinical research studies, and can be contacted for consultation by completing our Service Request Form.
What is source documentation?
Source documentation is the first documentation of the primary data point(s) for a study’s participant; for example, where the blood pressure measurement during a particular study visit was first recorded. The International Council for Harmonization Good Clinical Practice Guidelines (ICH GCP) define source documentation as “Original documents, data, and records (e.g., hospital records, clinical and office charts, laboratory notes, memoranda, subjects’ diaries or evaluation checklists, pharmacy dispensing records, recorded data from automated instruments, copies or transcriptions certified after verification as being accurate copies, microfiches, photographic negatives, microfilm or magnetic media, x-rays, subject files, and records kept at the pharmacy, at the laboratories and at medico-technical departments involved in the clinical trial)”.
Essentially, source documents are where data regarding study participants are first recorded. These documents and files can be paper or electronic; collected specifically for the study (e.g., height and weight) or part of a patient’s medical record (e.g., lab results). (Although it should be noted that often lab results are collected specifically for a study.) Sometimes data is entered directly into an electronic database, such as an online quality of life questionnaires, but this would still be considered source documentation. To keep track of all documentation, it is always helpful in clinical research to develop a document that outlines where your source data is located. This avoids confusion over similar, non-source data that might be located in different parts of a patient record. For example, in a study looking at a possible treatment for acute spinal cord injury, the time of injury may be important. Often this data is recorded in several different locations in the medical record. By documenting which location you are using for the study, there is no confusion for new staff, monitors, auditors, or Health Canada inspectors.
“The consequences of inadequate, inaccurate, or missing source documentation can range from mild to severe, which may result in a site or a study being shut down.”
Why is it important?
The main purpose of source documentation in clinical research is that it allows an independent person to be able to reconstruct the data collected in a study and ensure that it was captured correctly. A large and diverse amount of data is often collected in clinical research; proper documentation ensures the integrity of the data and “proves” that the trial respected ethics, participants’ well-being, and the study protocol. Per ICH GCP, all clinical research data should be recorded, handled, and stored in a way that allows its accurate reporting, interpretation, and verification. In the acute spinal cord injury example above, time of injury may be a critical inclusion criterion (i.e., a participant must be treated within 48 hrs of injury, and could lead to a participant being excluded unnecessarily). Alternatively, using the incorrect source data may lead to a participant being included when they were not eligible. A much more likely scenario is that staff would collect data inconsistently, leaving the monitor, auditor or inspector unable to verify the data.
Specific points to consider:
- Each data point in the case report form should be traceable to a source document
- Every interaction with a study participant should be documented
- Data should be collected and documented in real time
- The person collecting the data, or whomever performed the assessment, should initial and date their work
Why do I need this?
The United States Food and Drug Administration (U.S. FDA) introduced the concept of ALCOA in the 1990s, as a response to an increase in ‘lack of data integrity’ findings during routine inspections. While ALCOA doesn’t specifically feature in the US FDA regulations, the concepts do. Further items such as Consistent, Enduring and Available have been added over the years, but in general ALCOA is now referred to as ALCOA-C, or ALCOA+. Source documentation should meet the ALCOA-C standards for record keeping. Florence Healthcare provides a great infographic to explain these standards: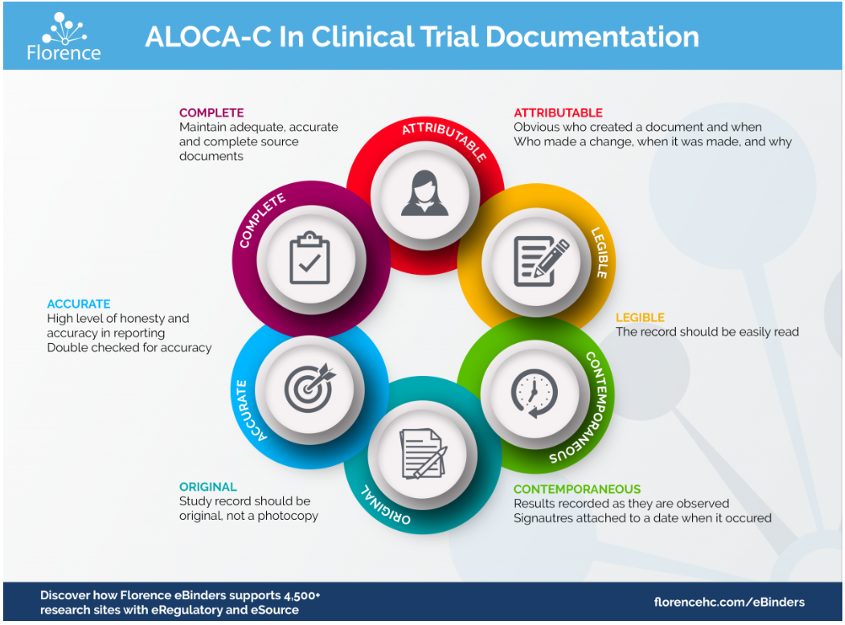
Inadequate, inaccurate, or missing source documentation is one of the most common findings in Health Canada inspections. In 2015–16, observations on record keeping were one of the top three observations cited under Health Canada regulation C.05.012 Records. This citation specifies that “the clinical trial records had errors and/or missing information that did not allow for complete and accurate reporting, interpretation and verification of the data”. The consequences of inadequate, inaccurate, or missing source documentation can range from mild (a data point cannot be validated, and may need to be left out of the study analysis) to severe (the majority of data cannot be traced to any source documentation during a Health Canada inspection) which may result in a site or a study being shut down.
In clinical research, the saying goes “If it isn’t documented, it didn’t happen”. Proper source documentation is vital to ensure that a study is legitimate, both in the accuracy of the results and during any follow-up inspection or additional analyses.
Links to references and resources
- GUIDELINE FOR GOOD CLINICAL PRACTICE (ich.org)
- ALCOA-C in Clinical Trial Electronic Document Management (florencehc.com)
- Inspectorate Program Annual Inspection Summary Report 2015 – 2016 – Canada.ca
- What is ALCOA+? Can it help Data Integrity? » The Good Practices Blog (laboratorypractice.com)
- Microsoft Word – Data Quality SQA Article 2007 ver.doc (southernsqa.org)



